Health
Cholera: Nigeria faces vaccine shortage as death toll hits 40
Cholera: Nigeria faces vaccine shortage as death toll hits 40
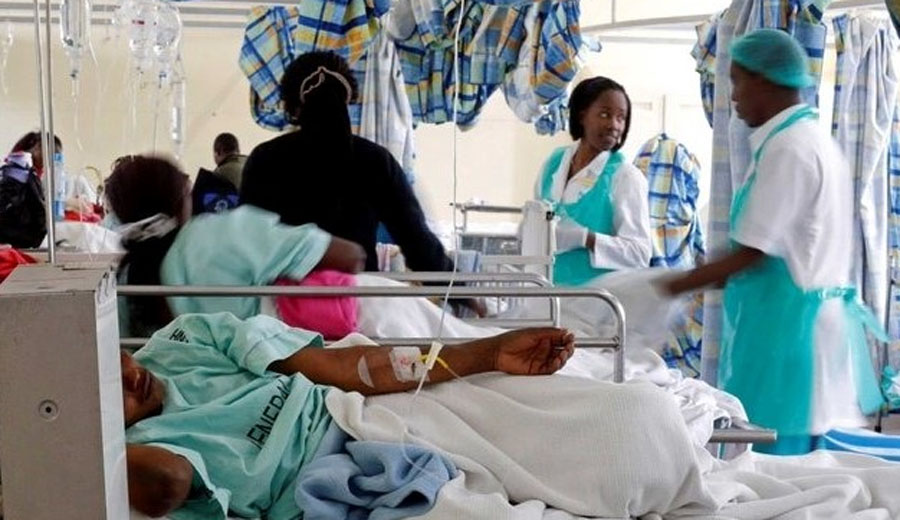
Nigeria – As the death toll from the latest cholera outbreak reaches 40, the Nigeria Centre for Disease Control (NCDC) has announced a critical shortage of vaccines. Dr. Jide Idris, Director General of the NCDC, confirmed that Nigeria has placed an order for additional cholera vaccines from donor agencies, but the delivery date remains uncertain.
Dr. Idris emphasized the importance of vaccines and other preventive measures to control the spread of cholera, a severe diarrhoeal disease caused by the ingestion of food or water contaminated with Vibrio cholerae. He noted, “We don’t have enough to prevent an outbreak because we need to give these things before that time. Most countries plan ahead. When it comes to health security, we are supposed to stockpile some things in anticipation of an emergency.”
Between 2021 and 2024, cholera has claimed 4,364 lives out of 139,730 suspected cases in Nigeria, according to NCDC reports. The rainy season has exacerbated the situation, with recent data showing an increase in cholera cases across the country. From January 1 to June 11, 2024, there were 1,141 suspected cases, 65 confirmed cases, and 30 deaths reported in 96 local government areas in 30 states.
READ ALSO:
- Kenya investigates police conduct in protests over new tax plan
- Peter Obi didn’t create Obidient Movement, says Aisha Yesufu
- Falana, other lawyers fault court ruling on Sanusi, Bayero dispute
The NCDC identified ten states contributing to 90% of the cholera burden: Bayelsa, Zamfara, Abia, Cross River, Bauchi, Delta, Katsina, Imo, Nasarawa, and Lagos. Lagos State has seen a significant rise in cases, with 417 suspected cases, 35 confirmed cases, and 24 deaths as of June 19, 2024. The cholera outbreak in Lagos was traced to an unregistered tiger nut drink, highlighting the role of contaminated food and beverages in spreading the disease.
Dr. Idris stressed the need for improved personal, environmental, and sanitary hygiene to prevent cholera. He also mentioned that while cholera vaccines are not long-lasting, a combination of vaccination and preventive measures is essential.
Public health experts, including Prof. Tanimola Akande and Dr. Kenechukwu Igwegbe, advocate for preventive vaccination against cholera, especially given its recurring nature in Nigeria. The World Health Organisation has identified three oral cholera vaccines – Dukoral®, Shanchol™, and Euvichol-Plus® – that require two doses for full protection.
The NCDC, along with other agencies, is actively working to manage the outbreak. However, Dr. Idris highlighted the challenges in vaccine procurement and the need for better planning and resources. “Cholera vaccine demand is far ahead of supply, so most people who need them place orders and plan ahead. It is the same with all vaccines, not just cholera.”
As Nigeria grapples with this cholera outbreak, the focus remains on obtaining more vaccines and enhancing preventive measures to curb the spread of this deadly disease. The NCDC, in collaboration with international and local partners, continues to provide support and resources to the affected states, aiming to mitigate the impact of this public health crisis.
Cholera: Nigeria faces vaccine shortage as death toll hits 40
Health
FG destroys another 200 containers of expired drugs

FG destroys another 200 containers of expired drugs
The Federal Government Joint Committee on the destruction of fake, illicit and expired pharmaceuticals has destroyed another 200 containers of substandard drugs worth billions of Naira imported into the country through the Apapa and Tin-Can Island seaports.
This is coming after the committee initially destroyed 250 containers of the same expired pharmaceutical products in January, 2025.
To this end, the committee comprising the Nigerian Customs Service (NCS), National Security Adviser (NSA), National Drug Law Enforcement Agency (NDLEA), National Agency for Food Drugs Administration and Control (NAFDAC) and Nigerian Army has destroyed a total of 450 containers of expired drugs in the first quarter of 2025.
The destruction exercise which took place in Epe, Lagos State yesterday, was part of a broader initiative of the Federal Government to curb importation of illicit and controlled drugs in order to safeguard public health and curb drug-related crimes in the country.
The pharmaceuticals destroyed were Tramadol, codeine, Apetamine, and Analgin injection amongst others.
Speaking to newsmen, the National Security Adviser (NSA), Mallam Nuhu Ribadu, said the initiative was important to prevent the circulation of dangerous substances such as Tramadol and Codeine, which are often abused, thus contributing to criminal activities in the country.
READ ALSO:
- Rivers court bars woman from answering ex-husband’s name
- PDP gives condition to back Atiku, Obi, El-Rufai in 2027
- Gov Fubara’s suspension dents Nigeria’s image, says ex-President Jonathan
Mallam Ribadu, who was represented by Ibrahim Sulaiman, said the destroyed drugs were seized at Apapa, Tin Can, and Lekki seaports.
He, however, disclosed that similar destruction was conducted in Port Harcourt as part of the first phase.
“The National Security Adviser is very passionate about ensuring that these illicit drugs do not remain in circulation. The government will not allow perpetrators to act with impunity; they will be arrested and prosecuted,” he stated.
The intercepted drugs were either expired or classified as controlled substances that should only be used under strict medical supervision.
Also, the Assistant Customs Controller, A.O Oguntuase from FOU Zone A, emphasised the enforcement efforts of security agencies in tracking and intercepting these illegal imports.
“Through intelligence gathering and swift action, we have been able to prevent the distribution of these drugs.
“The value of the destroyed substances is enormous, with each container estimated to be worth close to a billion naira,” he said.
On his part, representative of the NDLEA, DC Omotosho Solomon, stated that suspects arrested in connection with the goods were being tried in court.
Solomon warned importers engaged in the trafficking of illicit drugs and pharmaceuticals to desist from such acts or face the full process of law on illegal importation.
FG destroys another 200 containers of expired drugs
Health
1,319 died of Diphtheria in Nigeria, says NCDC
1,319 died of Diphtheria in Nigeria, says NCDC
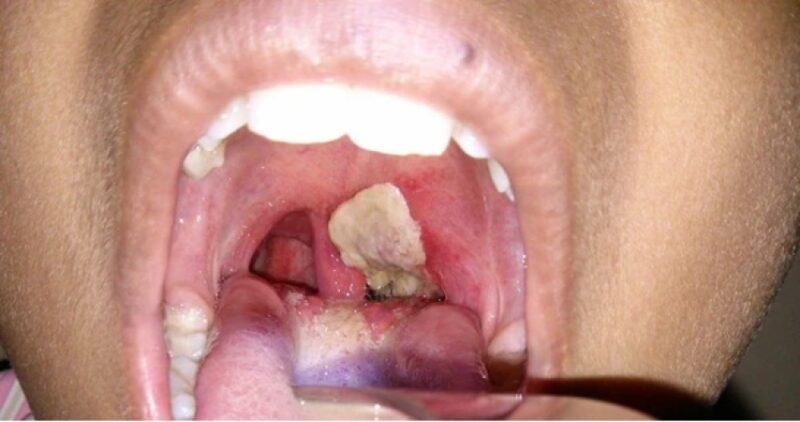
The Nigeria Centre for Disease Control and Prevention (NCDC) has confirmed that the ongoing diphtheria outbreak in the country has claimed 1,319 lives.
The alarming figure was disclosed in the latest situation report from the National Diphtheria Emergency Operations Centre (EOC), published on the agency’s official website.
Diphtheria, a highly contagious bacterial infection caused by Corynebacterium diphtheriae, primarily affects the mucous membranes of the throat and nose.
The bacteria produce a dangerous toxin that can cause severe complications such as breathing difficulties, heart failure, and nerve damage.
According to the NCDC, more than 42,000 suspected cases have been recorded across 37 states since 2022. The latest report covers epidemiological week 10 of 2025 and highlights ongoing challenges in controlling the outbreak, including low vaccination coverage, inadequate laboratory capacity, and delays in confirming cases,” the agency stated.
Kano State accounts for 75% of confirmed cases, with 18,108 out of 25,812 recorded in the country. Other high-burden states include Bauchi with 2,334 cases, Yobe with 2,408 cases, and Katsina with 1,501 cases.
READ ALSO:
- TUC backs NYSC member for criticising Tinubu administration
- Governors lobby Tinubu over direct allocation to LGAs
- Peace deal must bar Ukraine from Nato, Russian official says
Out of the 42,642 suspected cases reported, 25,812, or 60.5%, have been confirmed, including 396 laboratory-confirmed cases, 216 epidemiologically linked cases, and 25,200 clinically compatible cases. A total of 7,769 cases, representing 18.2%, have been discarded, while 3,591 cases, or 8.4%, remain pending classification. Another 5,470 cases, making up 12.8%, have an unknown classification.
The confirmed cases are spread across 184 Local Government Areas in 26 states. Kano, with 18,108 cases, has the highest number, followed by Bauchi with 2,334 cases, Yobe with 2,408 cases, Katsina with 1,501 cases, Borno with 1,161 cases, Jigawa with 53 cases, Plateau with 119 cases, and Kaduna with 44 cases. These states together account for 99.7% of all confirmed infections.
The NCDC reported that the overall case fatality rate stands at 5.1%, meaning that one in every 20 confirmed cases results in death. However, some states have recorded significantly higher mortality rates. Kaduna has a fatality rate of 58%, Plateau 48%, Lagos 83 per cent, and Adamawa 80%.
In the latest reporting week, 23 new suspected cases were identified, 20 in Lagos and three in Katsina. However, none have yet been confirmed, with 56.5% of cases still unclassified due to delays in laboratory testing.
To curb the outbreak, the NCDC stated that the federal government has launched reactive vaccination campaigns in high-burden states and intensified community engagement efforts, particularly through traditional and religious leaders.
The agency urged the public to ensure they are vaccinated and to seek medical attention immediately if they experience symptoms such as sore throat, fever, or difficulty breathing.
1,319 died of Diphtheria in Nigeria, says NCDC
Health
NAFDAC uncovers syndicate scamming foreign firms with fake documents

NAFDAC uncovers syndicate scamming foreign firms with fake documents
The National Agency for Food and Drug Administration and Control (NAFDAC) has uncovered a fraudulent syndicate impersonating the agency to scam foreign companies using fake documents.
The agency announced the discovery in a statement by Prof Mojisola Christianah Adeyeye FAS, its Director General, following a petition from Thani Almaeeni Trading Group, Abu Dhabi, UAE, which fell victim to the scam.
It was revealed that the syndicate is being led by one Ikoro Mang Ifendu who was arrested on February 7, 2025, in Aba, Abia State for allegedly defrauding foreign firms by posing as a NAFDAC official.
“Mr. Ikoro Mang Ifendu of 26, Park Road, Aba Abia State, born on the 2nd of January, 1973, was arrested on the 7th of February 2025 at Ogborn Hills, Aba, Abia State in a case of alleged fraud and obtaining huge funds by false pretence from unsuspecting foreigners that cuts across various countries as a NAFDAC Staff. He is a native of Amamba Village, Abiriba in Ohafia LGA, Abia State. His level of education is SSCE and he deals in clothing materials. The suspect is in our custody and is cooperating with the investigation,” the agency stated.
How the syndicate operated
NAFDAC disclosed that the syndicate used a sophisticated three-pronged scheme involving a buyer, a bank, and a lawyer. First, a buyer would contact foreign firms to purchase goods for importation into Nigeria.
READ ALSO:
- Naira strengthens, gains N29.89 against US dollar
- EL-Rufai, police clash over ex-commissioner’s arrest
- How to lower fuel consumption of your car
The company would then be introduced to a bank, where they were informed that NAFDAC approval was required for transactions. A lawyer would subsequently be introduced, claiming to facilitate NAFDAC certification for a fee. Fraudulent receipts and counterfeit NAFDAC certificates were issued to victims.
The fraudulent operation extended across multiple banks and accounts. Investigations revealed that the group managed 15 domiciliary and 5 local accounts in seven Nigerian banks, with an inflow of over $950,000 into Nigerian accounts and $450,000 into offshore accounts in Cotonou, Benin Republic. Even after Ifendu’s arrest, foreign victims continued to send money, with a recent alert of $75,000 received in one of the accounts.
Fake documents used in the scam
The documents used to deceive foreign companies included:
- A counterfeit Certificate of Registration for Dried Fish (Seafood) under the NAFDAC Registration No. A2-7059, issued to Thani Almaeeni Trading Group.
- Fake revenue receipts, including a N6,687,000 receipt dated November 28, 2024, and another N20,900,000 receipt dated December 13, 2024.
- A forged processing requirement document for $13,785, purportedly signed by a former NAFDAC director who retired a year earlier.
- Fraudulent Swift Advice copies, including a $1,990 transaction to Rosemary Obosi and a $3,000 transaction to Ifendu.
- A telegraphic transfer record showing USD 132,972.84 sent to Citycode Savings & Loans, Lagos, on December 24, 2024.
The fraudsters also used letterheads falsely to identify themselves as HALI & CO Chambers, operated by Ifendu and Obosi.
Foreign companies affected
- Aside from Thani Almaeeni Trading Group, other victims identified include
- Japan Long Tie (China) Co. Ltd, Jinan, China (importation of condoms
- BEYOND-Korea, USA
- Aquaforest SP, Poland (drinks and vegetable oil registration)
- Nomea srl, Italy
- Tianyan Filter Cloth Co. Ltd, China
- Siam Canadian China Ltd, China (frozen onion spices)
Case transferred to EFCC
Following NAFDAC’s findings, the case will be transferred to the Economic and Financial Crimes Commission (EFCC) for further investigation. The probe will extend beyond NAFDAC-regulated products to track assets acquired through fraudulent activities.
“This investigation will invoke POCA [Proceeds of Crime Act] on all the assets illegally acquired in the course of these fraudulent activities,” NAFDAC stated.
NAFDAC has warned businesses against engaging consulting firms claiming to facilitate product registrations.
The agency advised firms to verify all NAFDAC-related registrations through its official portal: https://registration.nafdac.gov.ng/
NAFDAC uncovers syndicate scamming foreign firms with fake documents
-

 metro2 days ago
metro2 days ago‘We’re not hiring,’ NNPC denies viral recruitment adverts
-

 metro1 day ago
metro1 day agoNatasha: Court blocks recall attempt, stops INEC
-

 Sports2 days ago
Sports2 days agoOdegbami speaks on Osimhen breaking his 44-year goals record
-

 metro3 days ago
metro3 days agoMore trouble brews in Rivers as Ijaw congress considers self-determination option
-

 Entertainment2 days ago
Entertainment2 days agoI didn’t snatch Asake’s mother from her husband -Musibau Alani
-

 Sports2 days ago
Sports2 days ago2026 WCQ: Super Eagles move up to third place with 2-0 win in Rwanda
-

 metro1 day ago
metro1 day agoBoko Haram attacks military base in Adamawa
-

 metro1 day ago
metro1 day agoOluwo accuses Ooni of plotting to dethrone him













