Health
Nigeria to get 20 million doses of COVID-19 vaccine
The Federal Government says it is expecting to receive 20 million doses of COVID-19 vaccine in the first quarter of 2021.
Executive Secretary, National Primary Health Care Development Agency, Faisal Shuaib, gave the indication at the presidential task force on COVID-19 media briefing in Abuja on Thursday.
He said Nigeria had put in place the machinery for this, adding that the country is a member of COVAX, an international coalition, under the WHO umbrella.
Shuaib added that 92 countries came together to ensure access and safety of vaccines.
He said the expected 20 million doses will first be given to workers in the health sector and vulnerable citizens.
“We are on course to access safe vaccine in the first quarter of 2021. We will be leveraging on the polio platform to ensure effective delivery of vaccines to our vulnerable population,” he said.
H added, “We have established a supra-ministerial advisory committee to ensure a seamless administration. A technical group meets every week and has devised a risk communication plan to deliver safe vaccines to Nigerians.”
A former Minister of Finance, Ngozi Okonjo-Iweala, has said Nigeria and other African countries will have access to COVID-19 vaccines as from the end of January through the first quarter of 2021.
A statement by the Ministry of Foreign Affairs said on Wednesday that Okonjo-Iweala disclosed this at a closed-door meeting with the Minister of Foreign Affairs, Geoffrey Onyeama in Abuja.
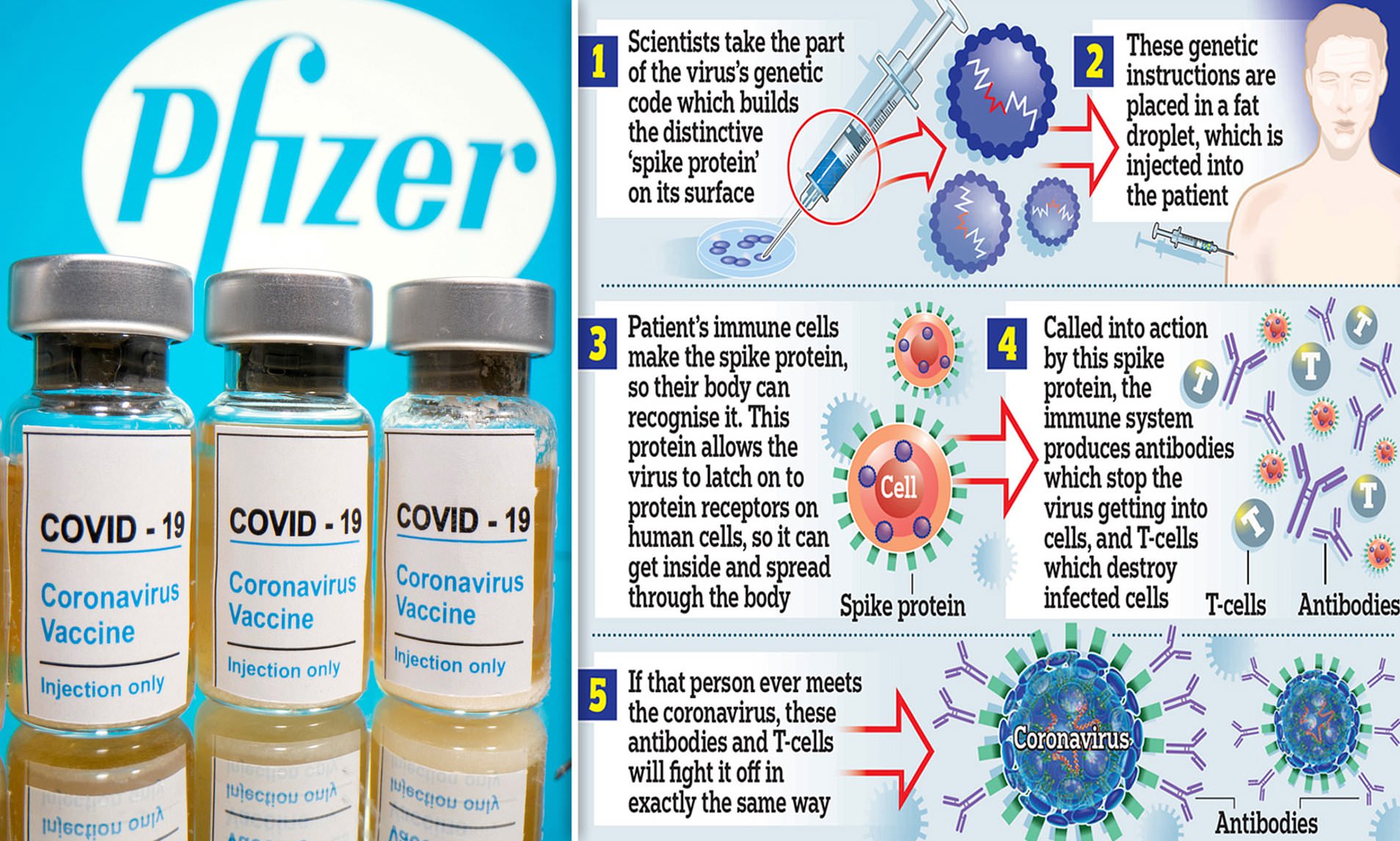
The big breakthrough came when Pfizer/BioNTech published its first results in November.
They showed the vaccine is up to 95 per cent effective and the UK is due to get 40 million doses.
The vaccine is given in two doses, three weeks apart. About 43,000 people have had the vaccine, with no safety issue.
The vaccine must be stored at a temperature of around -70C and transported in a special box, packed in dry ice and installed with GPS trackers.
On 2 December, the UK became the first country in the world to approve the Pfizer/BioNTech coronavirus vaccine for widespread use.
On 8 December, 90-year-old Margaret Keenan became the first patient to receive the vaccine at University Hospital in Coventry, with 800,000 more doses expected to be given in the coming weeks.
Health
Just In: Nigeria receives 846,000 malaria vaccine doses

Just In: Nigeria receives 846,000 malaria vaccine doses
Nigeria has received 846,000 doses of a groundbreaking malaria vaccine from development partners to reduce the country’s high incidence of the disease, especially among children and other vulnerable groups.
Prof. Muhammad Pate, Coordinating Minister of Health and Social Welfare, announced during the event on Thursday in Abuja that the vaccines were expected to play a pivotal role in lowering malaria-related deaths.
The News Agency of Nigeria (NAN) reports that large clinical trials in Africa proved the vaccine, RTS,S/AS01 (Mosquirix) as effective in reducing contracting malaria cases and mortality among young children.
Nigeria is the third African country to introduce the vaccine, following Ghana and Kenya, which began using it in 2023.
Malaria affects 97 per cent of Nigeria’s population. Its transmission occurs year-round in southern regions and lasts up to three months in the northern regions.
The primary malaria vectors are Anopheles coluzzii and Anopheles gambiae, with Anopheles funestus playing a secondary role in some areas.
The vaccines come as Nigeria intensifies efforts to reduce malaria-related deaths under the National Malaria Strategic Plan (NMSP) 2021–2025.
Pate said the vaccine would significantly boost the country’s ongoing malaria elimination efforts.
“Our target is to prioritize regions most affected by malaria, particularly in rural areas where access to healthcare is limited,” he said.
He also said the vaccine rollout would begin in high-burden regions before expanding nationwide.
According to him, global health partners, including WHO, UNICEF, and Gavi, are providing technical and financial support to ensure the successful implementation of the vaccine.
READ ALSO:
- Biden okays $4.5bn student debt relief as vote nears
- Don Jazzy raises Verydarkman NGO with N100m
- EFCC calls 15th witness in Fayose fraud trial
Just In: Nigeria receives 846,000 malaria vaccine doses
Health
Cholera ravages 28 LGs in Kano, kills 45
Cholera ravages 28 LGs in Kano, kills 45
The Coordinator of the Kano State Centre for Disease Control (KNCDC), Prof. Muhammad Adam Abbas, has revealed that a recent cholera outbreak has claimed 45 lives across 28 local government areas in the state.
He stated this on Tuesday at a retreat organised by the Kano State’s Ministry of Health in Kaduna.
According to Prof. Abbas, cholera thrives in environments with poor sanitary conditions, and maintaining high standards of personal and food hygiene is vital in combating its transmission.
READ ALSO:
- Police in Imo burst robbery gang terrorising Owerri
- Ahead Ondo poll, PDP demands removal of INEC commissioner
- Pro-Fubara lawmakers react to Amaewhule-led faction declaring their seats vacant
He advised that frequent handwashing, maintaining a clean environment, thoroughly washing fruits and vegetables and proper food storage are essential preventive measures.
He also highlighted the increased risk during the rainy season, as running water can wash contaminants, including dirt and faeces, into low-lying wells.
To mitigate this, he recommended building parapet embankments around wells to prevent contamination.
Prof. Abbas further noted that the government is taking proactive steps to control the outbreak, including chlorinating water sources in communities with poor sanitation and those affected by the disease.
Cholera ravages 28 LGs in Kano, kills 45
Health
Breakthrough in wound healing, electric suture creates excitement

Breakthrough in wound healing, electric suture creates excitement
A new type of suture that combines electrical stimulation with traditional suturing methods has been developed.
It has been demonstrated that this mechanoelectric suture, which is constructed from biodegradable materials and generates electric fields in response to movement, significantly accelerates wound healing and lowers the risk of infection.
This innovative approach could revolutionise wound closure procedures and offer patients a faster and safer healing process.
In an experiment in rats, the new strong, flexible thread hastened wound healing by transforming muscle movement into electricity, researchers report in Nature Communications.
According to materials scientist Chengyi Hou of Shanghai’s Donghua University, the substance “could change how we treat injuries” if it is finally shown to be safe for use in humans.
READ ALSO:
- Rumour of Mohbad’s widow Wunmi expecting another child sparks shocking reactions
- Bobrisky: I was treated badly during my visit to EFCC office, says Verydarkman (VIDEO)
- Kano: Lebanese jailed 14 years for sexually exploiting two teenage girls
Although prior methods depended on large external batteries, researchers already knew that applying electricity to sutures helped accelerate the healing process. The body provides the energy for the new sutures.
The thread is composed of magnesium, a metal that the body may eventually absorb, and biodegradable polymers. The central layer of the thread rubs against the outer shell when the muscles around the sutures contract and relax, which transfers electrons to the shell and produces electricity.
Hou and colleagues used the thread’s electrical stimulation to treat artificial wounds in lab dishes. After 24 hours, fibroblasts, which are critical to healing, reduced the wound area from 69 percent to 11 percent. Untreated artificial wounds reduced in size from 69 percent to 33 percent after 24 hours.
Rodents treated with electrical sutures recovered faster and were less likely to acquire infections than rats treated with regular sutures or left untreated. Next, the team intends to try the sutures on bigger animals.
Breakthrough in wound healing, electric suture create excitement
-

 News2 days ago
News2 days agoFIRS announces recruitment of young graduates
-

 metro3 days ago
metro3 days agoUpdated: Army says reports of COAS Lagbaja’s death fake news
-

 metro2 days ago
metro2 days agoOluwo chased me out when I visited him – Ooni (VIDEO)
-

 News2 days ago
News2 days agoOsinbajo traitor, can’t talk about integrity – Reno Omokri
-

 metro2 days ago
metro2 days agoFani-Kayode: How my aide died in hotel after attending church
-

 Business1 day ago
Business1 day agoWe’re settling out of court with NNPC, others — Dangote
-

 metro2 days ago
metro2 days agoBREAKING : DSS replaces Tinubu’s chief security officer
-

 News2 days ago
News2 days agoYou can’t overrule Supreme Court on LG autonomy – Klinsmann tells Soludo








You must be logged in to post a comment Login