Health
BREAKING: 90-year-old gets first COVID-19 vaccine in UK
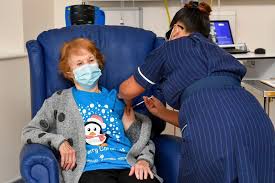
A COVID-19 vaccine has been injected into a patient for the first time in the UK.
ITV News reports that Margaret Keenan, who is from Coventry and will be 91 next week, said, “I feel so privileged to be the first person vaccinated against COVID-19.
“It’s the best early birthday present I could wish for because it means I can finally look forward to spending time with my family and friends in the New Year after being on my own for most of the year.”
The jab, made by US pharmaceutical firm Pfizer and German manufacturers BioNTech, will continue to be rolled out in 14 locations across the UK on Tuesday as the country’s biggest vaccination program kicks off.
And fronting the start of the fight back against the coronavirus pandemic is retired jeweller Margaret, who wore a blue Christmas t-shirt as the jab was quickly given without her “feeling a thing.”
With three weeks until her second jab, she should complete her course of injections just after Christmas, with full immunization expected in the first week of January.
Husband and wife Hari and Ranjan Shukla are also set for their first doses on Tuesday in Newcastle and said they feel “the crisis is going to come to an end.”
Shukla, who said he got the call on Friday from his doctor, said on Monday: “It’s an excellent idea, we will certainly do whatever we can because we are very excited about it.
“When we heard that we’ve got the vaccine now, we felt that the crisis is going to come to an end.”
He added, “I was very excited I got the opportunity of joining in and taking part, so we are very, very pleased and happy and excited as well.”
The couple join Margaret among the few to be vaccinated before the New Year, with the government expecting “the majority” of vulnerable people to follow suit in January and February.
Downing Street would not confirm whether they were expecting all 4 million doses of the Pfizer vaccine to arrive by the end of the year.
Croydon University Hospital in south London was one of the first hospitals to take delivery of the vaccine over the weekend, with similar scenes unfolding around the country ahead of the rollout.
Around 25 million people are covered by the 10 priority categories set out by the Joint Committee on Vaccination and Immunisation.
The first vaccinations will go to care home staff and residents, NHS frontline workers and people aged 80 and over – around six million people.
But Westminster stressed that two further vaccines were still being assessed by regulators, which could boost the number of doses available.
There are 800,000 doses in the first tranche, meaning 400,000 people will be vaccinated initially.
![]()
Health
Health Experts Warn: Everyday Habits Quietly Damaging Kidneys
Health Experts Warn: Everyday Habits Quietly Damaging Kidneys
Medical experts have raised fresh concerns over a growing number of lifestyle habits that may be silently damaging the kidneys, urging the public to adopt healthier routines to prevent long-term complications.
The warning comes amid increasing cases of kidney-related illnesses, many of which are linked to preventable behaviors such as poor diet, lack of exercise, and inadequate sleep.
According to health professionals, one of the leading contributors to kidney damage is unmanaged high blood pressure. Persistent hypertension places strain on the kidneys’ delicate filtration system, reducing their ability to function effectively. If left untreated, it may also contribute to the formation of kidney stones, which can cause severe pain and other complications.
Dietary choices have also come under scrutiny. Experts note that the rising dependence on processed and ready-made meals—often high in salt and phosphorus—significantly increases the risk of kidney disease over time. They recommend home-cooked meals using fresh ingredients as a healthier alternative.
Sleep patterns, often overlooked, play a critical role in maintaining kidney health. Studies indicate that poor or insufficient sleep disrupts the body’s internal clock, which is closely linked to kidney function. Over time, this disruption may lead to early signs of renal impairment.
Smoking remains another major risk factor. Beyond its well-known impact on lung health, tobacco use has been associated with an increased risk of kidney cancer and reduced blood flow to vital organs, including the kidneys. This reduced circulation can impair their ability to filter waste from the body.
Similarly, excessive alcohol consumption has been identified as harmful. While moderate intake may not pose significant risks, heavy drinking can lead to dehydration and elevated blood pressure—both of which negatively affect kidney performance.
A sedentary lifestyle is also contributing to the problem. Health experts explain that lack of physical activity increases the risk of obesity and metabolic disorders, conditions that are closely tied to kidney disease. Regular exercise, they say, is essential for maintaining overall organ health.
Additionally, nutritionists caution against excessive consumption of animal protein. While protein is necessary for body functions, high intake—particularly from animal sources—can increase acid production in the body. This forces the kidneys to work harder to maintain balance, potentially accelerating their decline over time.
Health authorities emphasize that kidney disease often develops gradually and without obvious symptoms, making prevention and early lifestyle changes crucial. They advise regular medical check-ups, balanced diets, adequate sleep, and physical activity as key measures to safeguard kidney health.
As awareness grows, experts hope that individuals will take proactive steps to reduce their risk and protect one of the body’s most vital organs.
Health Experts Warn: Everyday Habits Quietly Damaging Kidneys
![]()
Health
Resident Doctors Suspend Strike, Demand Payment of 19 Months’ Allowances

Resident Doctors Suspend Strike, Demand Payment of 19 Months’ Allowances
The Nigerian Association of Resident Doctors (NARD) has suspended its planned total and indefinite strike following renewed commitments from the Federal Government on salary payments, allowances, and welfare-related issues affecting medical professionals across Nigeria.
The decision was reached after an emergency virtual meeting of NARD’s National Executive Council (NEC) held on Saturday, where members reviewed ongoing negotiations with government institutions. The resolution was contained in a communiqué signed by NARD President Dr. Mohammad Usman Suleiman, Secretary-General Dr. Shuaibu Ibrahim, and Publicity and Social Secretary Dr. Abdulmajid Yahya Ibrahim.
According to the association, the suspension followed engagements with key government stakeholders, including the Vice President’s office, the ministries of health, labour, and finance, the Budget Office, the Office of the Accountant-General of the Federation, and other relevant agencies involved in health sector financing and administration.
NARD noted that there had been renewed assurances from the government regarding the implementation of the reviewed Professional Allowance Table (PAT), which is expected to reflect in April salary payments and beyond. The association also confirmed a reversal of earlier delays in implementation.
READ ALSO:
- Dangote Sugar Plans ₦485.9bn Rights Issue for Expansion Drive
- Phyna Says She Prefers Raising Pets Over Having Children
- Man City Fight Back to Beat Southampton, Reach Record FA Cup Final
Among its key demands, the association insisted on the immediate payment of all outstanding promotion arrears and salary backlogs, as well as urgent settlement of 19 months’ unpaid professional allowance arrears owed to members across various federal health institutions.
The doctors also called for the expedited release of funds under the 2026 Medical Residency Training Fund (MRTF), stating that approval has already been secured and disbursement processes are expected to be completed without further delay.
Another major concern raised by the association is the continued delay in payment of house officers’ salaries, which it described as a recurring issue requiring urgent intervention. NARD urged the authorities to convene an emergency stakeholders’ meeting to resolve the matter and ensure timely remuneration of newly qualified doctors.
While the strike has been suspended, the association stressed that its demands remain in force and warned that progress will be closely monitored. It said the situation will be reviewed at its upcoming May Ordinary General Meeting in Kano, where further action could be decided depending on government compliance.
Healthcare analysts note that the suspension offers temporary relief to Nigeria’s already strained health system, which has faced repeated disruptions due to industrial disputes over welfare and funding challenges.
Resident Doctors Suspend Strike, Demand Payment of 19 Months’ Allowances
![]()
Health
NAFDAC Busts Fake Alcohol Factories in Lagos, Seizes ₦350m Counterfeit Drinks

NAFDAC Busts Fake Alcohol Factories in Lagos, Seizes ₦350m Counterfeit Drinks
The National Agency for Food and Drug Administration and Control (NAFDAC) has uncovered and dismantled two major illegal production sites involved in the counterfeiting and adulteration of alcoholic beverages in Lagos State, seizing fake drinks valued at about ₦350 million.
Acting on credible intelligence, enforcement operatives carried out coordinated raids at Zamfara Plaza in the Trade Fair Complex and on Lagos Island, where large-scale production and packaging of counterfeit alcoholic beverages were taking place.
At the Trade Fair Complex, officials discovered a fully organised illegal production setup equipped with plastic mixing tanks, improvised filtration systems, empty branded bottles, corks, and packaging materials used to mimic popular alcoholic brands. The facility was allegedly used to manufacture and distribute fake drinks to unsuspecting consumers across Lagos.
A second raid on Lagos Island led to the arrest of a suspect linked to the illegal production and distribution network operating through a retail outlet. Authorities confirmed that all recovered items were seized and evacuated for laboratory analysis and further regulatory action.
READ ALSO:
- Senator Barau Empowers APC Leaders in Kano with 47 Cars, 282 Motorcycles
- Haaland Fires Man City Top as Burnley Relegated After 1–0 Defeat
- FBI Hunts Nigerian Woman in Multi-Million-Dollar Romance Fraud Case
According to NAFDAC, more than 1,800 cartons of counterfeit and adulterated alcoholic products were recovered during the operation, marking one of the agency’s significant recent enforcement breakthroughs in Lagos.
The agency warned that consumption of such adulterated alcoholic beverages poses severe public health risks, including poisoning, liver damage, kidney failure, and potentially death. It stressed that many of the products are produced under unhygienic and unsafe conditions without regulatory approval.
NAFDAC reiterated that the crackdown is part of a sustained nationwide campaign against food and drug counterfeiting, aimed at protecting public health and sanitising the supply chain for consumable goods in Nigeria.
The agency also warned producers, distributors, and retailers involved in such illegal activities to desist immediately, stating that offenders will face prosecution under existing food and drug safety laws.
Members of the public were urged to remain vigilant when purchasing alcoholic beverages, verify product authenticity, and report suspicious activities to the nearest NAFDAC office or through its toll-free line 0800-1-NAFDAC (0800-1-623322).
NAFDAC Busts Fake Alcohol Factories in Lagos, Seizes ₦350m Counterfeit Drinks
![]()
-

 metro3 days ago
metro3 days agoViral Threesome Video: Sisi Alagbo’s Husband Accepts Responsibility, Seeks Forgiveness
-

 Entertainment2 days ago
Entertainment2 days agoJubilation as Lateef Adedimeji, Mo Bimpe Welcome Triplet Boys
-

 Sports1 day ago
Sports1 day agoCarter Efe Defeats Portable in Celebrity Boxing Showdown
-

 News3 days ago
News3 days agoTinubu Nominates Tegbe as Minister of Power, Seeks Senate Confirmation
-

 metro2 days ago
metro2 days ago“No Calls, No Love”: Woman Shares Pain of Being Single at 50
-

 News2 days ago
News2 days agoSanwo-Olu Approves N50,000 Wage Award for Lagos Workers
-

 metro2 days ago
metro2 days agoTeen Murder Suspect Confesses, Reveals Chilling Details in Ikorodu Killing
-

 metro22 hours ago
metro22 hours agoOsogbo United Youth Forum Calls for Peace, Facts in Boundary Dispute, Rejects Divisive Claims
















You must be logged in to post a comment Login